Understanding droplet junction designs in microfluidics: From T-junctions to double emulsions and multi-reagent systems
Droplet microfluidics relies on junction design to control how droplets form, evolve, and perform. From simple T-junctions to advanced multi-reagent and double-emulsion systems, each geometry offers distinct advantages in size control, stability, and scalability. This guide breaks down key designs and helps match them to real-world applications and production goals.
Droplet microfluidics enables the generation of highly uniform droplets that function as microreactors, particle templates, and encapsulation carriers. By precisely controlling fluid interactions in microscale channels, researchers can tailor droplet size, composition, and internal structure with exceptional reproducibility. This level of control underpins applications in drug delivery, advanced materials, diagnostics, and cell biology.
At the core of droplet generation is the junction, where immiscible fluids meet and break into droplets. Junction geometry, the number of junctions in a device, and the number of reagents introduced together determine the droplet type and structural complexity.
When selecting a microfluidic platform, a practical question often arises:
Which junction geometry should you choose—and why?
The answer depends on your materials, performance targets, and production goals. Below, we outline the main junction designs and share practical insights we give to new team members and collaborators.
Fundamental droplet junction geometries
1. T-junction
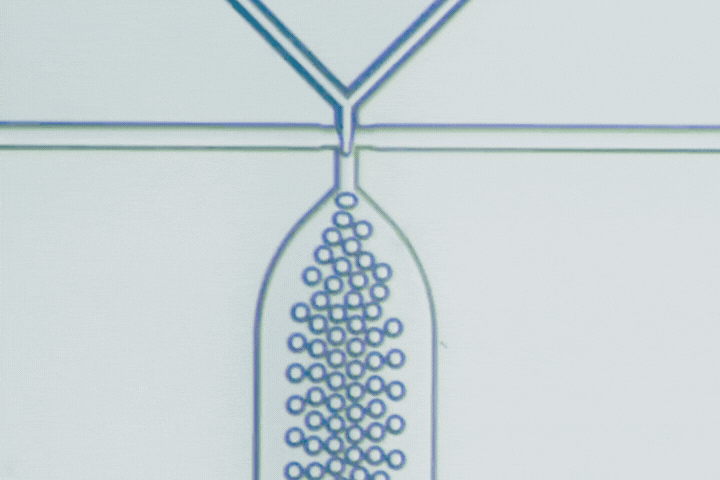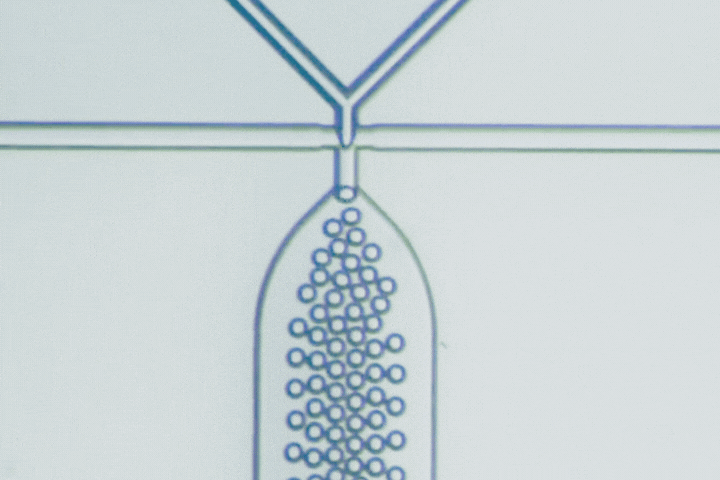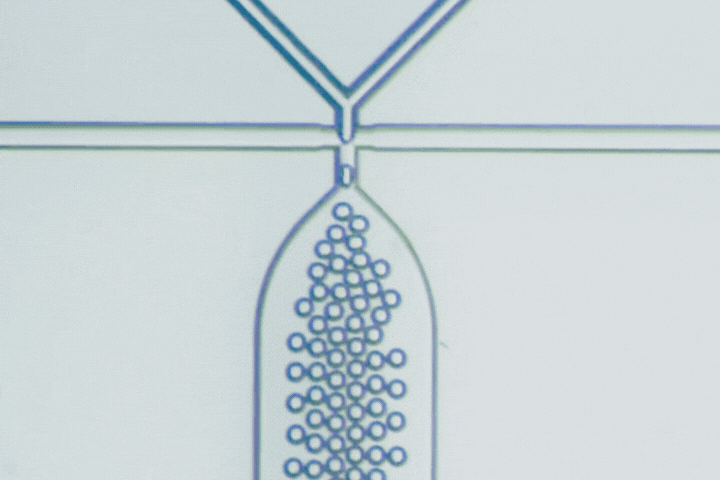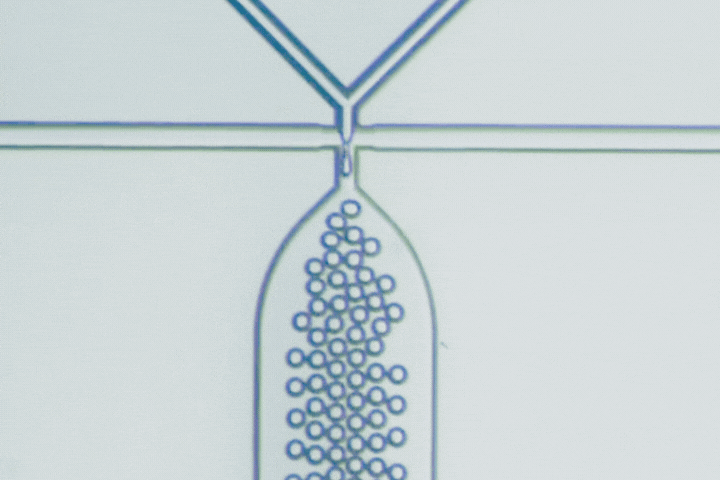
The T-junction is the most straightforward droplet generator. The dispersed phase enters a main channel carrying the continuous phase at a right angle. Shear forces from the continuous flow pinch the dispersed stream into droplets.
Droplet size is primarily governed by the balance between viscous shear and interfacial tension. Adjusting the flow-rate ratio provides a simple way to tune droplet diameter and production frequency.

Beyond conventional droplets, T-junctions can operate in a slug flow regime, intentionally forming elongated droplets larger than the channel width. This is useful when larger reaction volumes, stronger internal mixing, or segmented-flow chemistry are needed.
Advantages
- Simple, robust design
- Easy tofabricate and operate
- Stable for routine experiments
Best suited for
- Droplets in the tens to hundreds of microns
- Early prototyping and feasibility studies
- Slug-based reaction workflows
Limitations
- Less precise for very small droplets
- Broader size distributions than advanced geometries
Practicalguidance:
New to droplet microfluidics? Start with T-junctions to quickly grasp how flow ratio, viscosity, and interfacial tension shape droplet formation — the foundation for all other geometries.
2. Flow-focusing junction
Flow-focusing offers tighter control over droplet size and uniformity. The dispersed phase flows through a central channel while the continuous phase squeezes from both sides. At a narrow orifice, the dispersed stream is compressed and breaks into droplets.
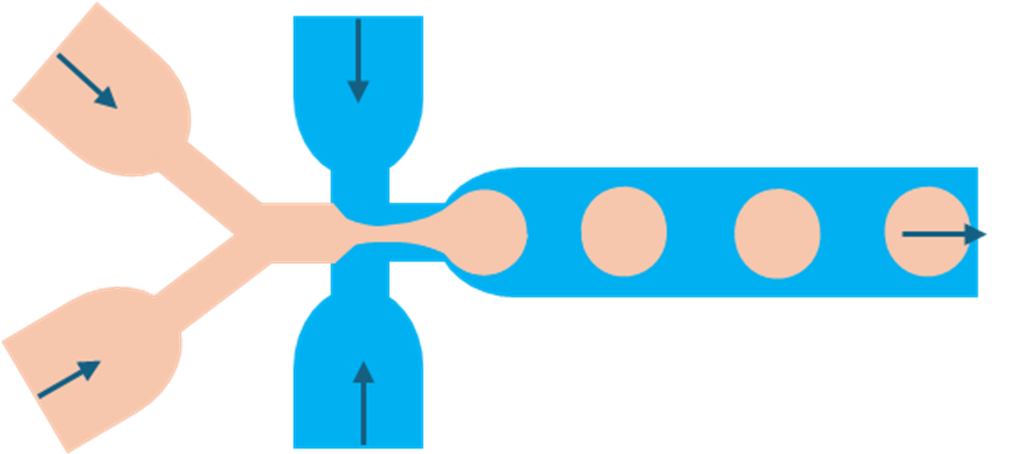
Because breakup occurs at a defined constriction, droplets are typically smaller and more monodisperse than those from T-junctions.
In practice, droplet size can often be tuned relative to junction size, meaning the droplet diameter is usually close to the channel width where droplets form:
- Low-viscosity systems (e.g., PLGA in organic solvent): droplet size can only vary within a narrow range, typically about ±10% of the junction width. Example: a 100 µm junction generally produces droplets around 90–110 µm.
- Higher-viscosity systems (e.g., alginate solutions): droplet size can be adjusted over a wider range, typically about ±30% of the junction width. Example: a 100 µm junction can produce droplets roughly 70–130 µm.
This difference occurs because higher-viscosity fluids resist breakup more strongly, allowing flow conditions to influence droplet size over a broader range.
Advantages
- Excellent size control
- High monodispersity
- Stable and reproducible formation
- Scalable by parallelization
Best suited for
- Tight size control
- Microparticle and nanoparticle production
- Droplet-based assays.
Limitations
- Sensitive to flow fluctuations
- Higher clogging risk
- Scale-up challenges
Example from our work:
Flow-focusing is widely used for PLGA microparticle production. Polymer solution droplets form first, then solidify through solvent diffusion and evaporation, yielding uniform particles for drug delivery and materials research.
Practical guidance:
Need tight particle size distribution? Flow-focusing is typically the go-to solution for precision and uniformity.
3. Pore-based junction
Pore-structured systems modify the outlet region of a flow-focusing junction by incorporating engineered pore features, rather than relying on a simple planar channel geometry. These pores reshape how fluids detach and move through the junction.
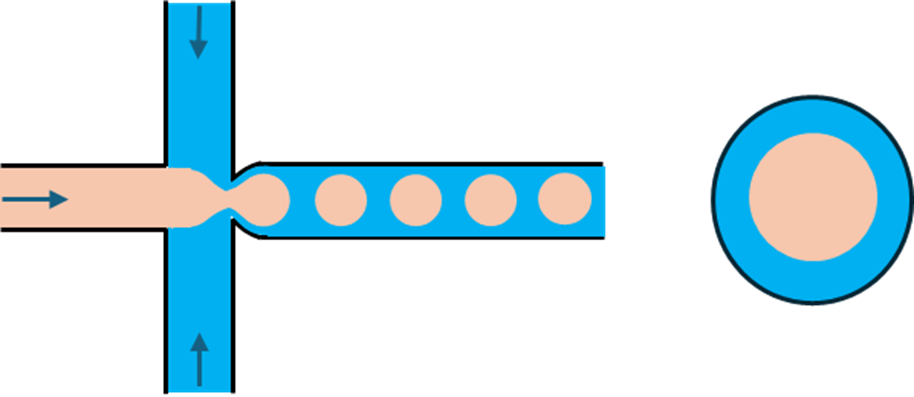
In this design, the pore structure on the outlet side induces the dispersed phase to detach from the top and bottom channel walls well upstream of droplet breakoff. This shortens the fluid–wall contact length and reduces areas where material can accumulate.
This approach is particularly valuable for polymer solution systems, such as PLGA microparticle production. Polymer streams often cause surface fouling in conventional flow-focusing chips, especially during solvent extraction or polymer precipitation. By reducing fluid contact with channel walls, pore-structured junctions lower polymer deposition and significantly reduce the risk of chip blockage. As a result, pore-structured flow-focusing devices enable longer continuous production runs, improved operational stability, and extended chip lifetime when working with sticky or fouling-prone materials.

Advantages
- Reduced clogging risk
- Stable operation with sticky or precipitating systems
- Longer continuous run times
Best suited for
- Polymer microparticle synthesis (e.g., PLGA)
- Long-duration continuous production
- Applications requiring robust and stable operation
Limitations
- More complex chip fabrication
- Harder to visually monitor droplet formation inside pores
Practical guidance:
Working with polymers? Conventional junctions can clog during precipitation. Pore-based designs enhance mass transfer and keep production running smoothly.
4. Step emulsification
Step emulsifications rely primarily on interfacial tension rather than shear forces. The dispersed phase passes through a small opening into a larger chamber, where surface tension causes predictable droplet pinch-off.

Because droplet formation is geometry-driven, these devices are less sensitive to flow fluctuations and can operate stably for long periods.
Advantages
- Highly stable droplet formation
- Minimal flow optimization
- Strong scalability through parallelization
Best suited for
- High-throughput production
- Manufacturing workflows
Limitations
- Limited droplet size tunability
- More complex chip design
- Lower flexibility for rapid prototyping
Practical guidance:
Scaling up production? If throughput and manufacturing stability matter more than flexibility, step emulsification is the smart choice. Learn more about scale-up strategies from Blacksheep Sciences.
5. Single vs Double junction systems
5.1. Single junction systems
Single breakup points generate single emulsions such as oil-in-water (O/W) or water-in-oil (W/O). These systems are simple and widely used in particle synthesis.
Example:
In agarose microparticle production, heated agarose solution forms droplets that gel upon cooling. Each droplet becomes a uniform hydrogel bead, with size determined by junction size.

5.2. Double junction systems
More complex structures can be produced using two sequential junctions. In these systems, droplets formed at the first junction are encapsulated within another fluid at a second junction, creating double emulsions.
Common examples include:
- Water-in-oil-in-water (W/O/W)
- Oil-in-water-in-oil (O/W/O)

These produce core–shell structures for controlled release, sensitive cargo encapsulation, and multi-stage reactions.
Challenges
- Requires precise flow balance
- Sensitive to surfactant chemistry
- Instability can cause shell rupture.
Practical guidance:
Keep it simple when possible — more complex structures demand tighter operational control.
6. Single vs multi-reagent configurations
6.1. Single reagent systems
A single dispersed stream creates droplets with uniform composition.
Example:
Polyacrylamide (PAM) beads form by polymerizing droplets containing monomer, crosslinker, and initiator. All components are combined off-chip and fed into a microfluidic chip as a single stream. Droplet size directly determines bead diameter and mechanical properties.

6.1. Multi-reagent systems
More advanced microfluidic devices introduce two or more reagent streams before droplet formation. These streams may merge within the channel and mix inside the droplets after formation.
Example:
Single-cell encapsulation co-flows cells and assay reagents, isolating individual cells for sequencing and high-throughput screening.
6.2. Janus droplets
An interesting variation occurs when two immiscible reagent streams enter the droplet generator side by side. Instead of fully mixing, they remain separated within the droplet, producing Janus droplets.
Janus droplets contain two distinct compartments with different compositions. This asymmetry enables unique functionality, such as particles with directional properties or dual chemical environments.
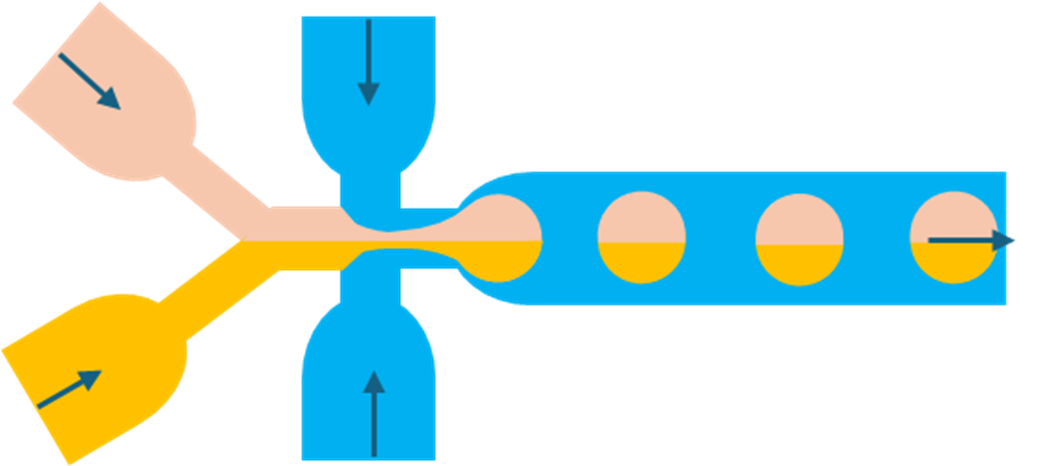
Practical considerations in droplet design
Selecting a droplet system depends on:
- Target droplet size
- Required monodispersity
- Number of reagents
- Droplet structure (single, double, or multi-compartment)
- Sensitivity of the materials involved
- Throughput needs
Typical choices:
- Flow-focusing for tight particle size control.
- Step emulsification for scalable hydrogel production.
- Multi-junction designs for stable double emulsions.
- Pore-based chip for polymer systems.
Successful droplet generation is not about geometry alone. It requires aligning chip design, flow control, and material compatibility.
From design to application
At Blacksheep Sciences, we help partners choose the right droplet architecture for their materials and production goals. Our team integrates chip engineering, materials science, and process optimization to translate lab concepts into robust workflows.
From PLGA drug-delivery particles to hydrogel materials and biological assays, we design droplet systems that are not only scientifically sound—but scalable, reliable, and production-ready.