PLGA-based drug encapsulation: Transforming delivery with microfluidics
Microfluidics is redefining PLGA-based drug encapsulation. While PLGA microspheres already enable controlled, long-acting therapies, microfluidics brings unmatched precision, consistency, and scalability. This powerful combination protects fragile drugs, enhances stability, and ensures targeted, sustained release - unlocking safer, more effective treatments for cancer, chronic disease, and beyond.
Delivering a drug exactly when and where it’s needed in the body is no longer just a laboratory vision - it’s already improving lives. One striking example is Risperdal Consta®, a long-acting injectable form of the antipsychotic risperidone. Oral risperidone is effective for schizophrenia and bipolar disorder, but daily dosing often leads to poor adherence, relapses, and repeated hospitalizations. By encapsulating risperidone in biodegradable PLGA (poly (lactic-co-glycolic acid)) microspheres, Risperdal Consta® enables controlled release over two weeks, maintaining steady therapeutic levels and reducing relapse risk - an FDA-approved proof that PLGA delivery can transform patient outcomes.
Now, microfluidics is taking PLGA encapsulation a step further. Traditional fabrication methods to make PLGA particles often lack precision. Microfluidics solves this by producing highly uniform particles at microscale, improving consistency, scalability, and performance. The biocompatibility and tunable release of PLGA combined with manufacturing precision provided by microfluidics are opening new frontiers in targeted therapy - from cancer drugs to nucleic acid delivery - with greater control than ever before.
Why encapsulation matters in drug delivery
An important approach in effective drug delivery is encapsulation - the process of wrapping an API (active pharmaceutical ingredient) within a carrier system. This approach protects sensitive compounds, enhances stability, improves solubility, and enables controlled release to specific targets in the body.
Encapsulation is especially critical for:
- Hydrophobic drugs like paclitaxel (a chemotherapy medicine) have low solubility in aqueous environments, which hinders absorption and reduces therapeutic impact.
- Hydrophilic APIs such as insulin (used in diabetes treatment) or gentamicin (antibiotic) can degrade quickly in the body, reducing treatment efficiency and necessitating frequent dosing.
- High toxicity compounds like doxorubicin (chemotherapy medicine) may affect healthy tissues as well as the diseased target tissue, leading to severe side effects.
- Drugs with low bioavailability such as cisplatin (chemotherapy medicine) are often cleared from the body before they reach their intended target, requiring higher doses to compensate, which may increase toxicity risks.
By optimizing how drugs are delivered, encapsulation technology can dramatically improve treatment outcomes. This is why microfluidics encapsulation methods - offering superior reproducibility and scalability over bulk mixing techniques – combined with PLGA, represent a game changing pair in the field of drug delivery.
Microfluidics: A leap forward in drug encapsulation
Microfluidics is the science of controlling tiny volumes of fluids - often smaller than a raindrop - through micro-scale channels on chips the size of a fingernail. These “lab-on-a-chip” platforms serve as miniature laboratories, enabling precise control over chemical and biological processes.
Microfluidic technology is transforming drug encapsulation and drug screening through:
- Precision engineering: microfluidic systems produce highly monodisperse particles with uniform size and shape to ensure predictable drug release and performance.
- Process efficiency: uses small fluid volumes and rapid mixing to minimize waste and accelerate production compared to traditional batch methods.
- Versatility: enabling advanced encapsulation formats - including single emulsions, double emulsions, and core-shell structures - suitable for a wide variety of drug types.

- Scalable design: microfluidic technology is increasingly designed to scale, making it possible to transition smoothly from laboratory development to industrial manufacturing.
This level of precision and flexibility makes microfluidics a foundational technology in the development of the next generation of drug delivery systems.
How PLGA-based encapsulation works with microfluidics
PLGA is a biodegradable, FDA-approved polymer that safely breaks down in the body through hydrolysis into lactic acid and glycolic acid, which are naturally metabolized and excreted. Thanks to its excellent biocompatibility and adjustable degradation profile, PLGA has become a trusted material for a range of medical applications, including sutures, implants, and drug delivery systems.
Crucially, PLGA can encapsulate both hydrophilic and hydrophobic APIs, making it ideal for controlled microfluidic-based drug delivery systems. When paired with microfluidic technology, this potential is further enhanced.
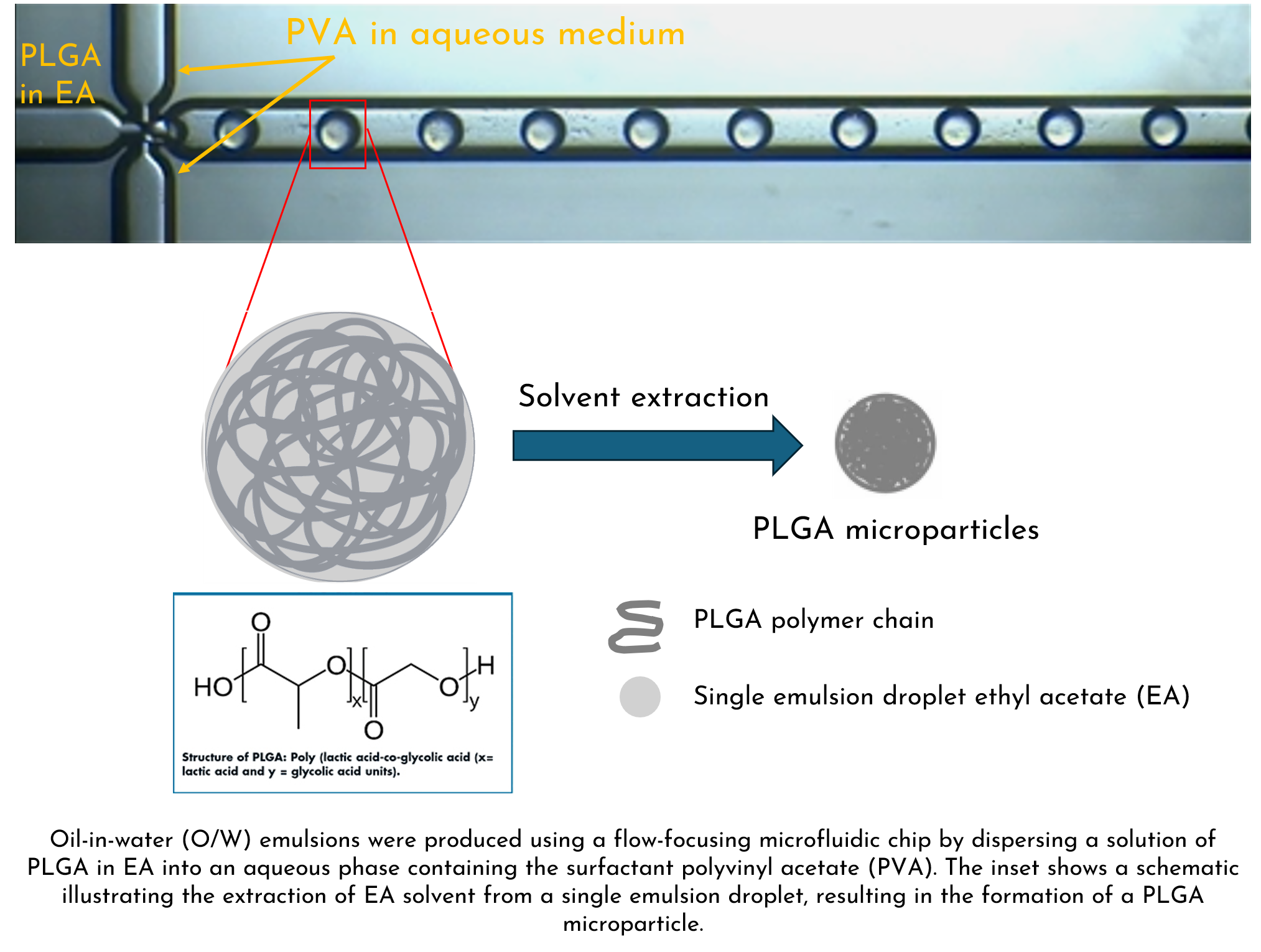
In a typical microfluidic process, PLGA and the drug are dissolved in a water-immiscible organic solvent like dichloromethane or ethyl acetate. This organic phase is then introduced into a microfluidic chip along with an aqueous phase. The chip’s flow-focusing design generates highly uniform emulsion droplets. As the solvent evaporates, solid PLGA microparticles form – each precisely loaded with the drug.
This approach enables the production of drug-loaded PLGA particles with finely tuned sizes, surface properties, and release profiles—offering control and reproducibility that far exceed conventional bulk methods.
Key advantages include:
- Precisely sized particles (typically 1-100 µm)
- Consistent drug encapsulation
- Low batch-to-batch variability
- Higher efficiency and yield
Such a platform is especially well-suited for delivering sensitive and costly therapeutics, including anticancer agents, peptides, and other biologics.
PLGA microfluidic encapsulation in action: hydrophobic vs. hydrophilic drugs
Hydrophobic drugs
Curcumin, the primary active compound in turmeric, is renowned for its potent anti-inflammatory and antioxidant properties, yet is limited by poor water solubility. In a single-emulsion microfluidic method, PLGA and curcumin were dissolved in ethyl acetate (a green solvent) and emulsified into an aqueous phase. The resulting uniform PLGA-curcumin microparticles (~46 µm, CV 3.8%) significantly enhance curcumin's solubility, protect the drug from degradation, and enable sustained drug release.

Hydrophilic drugs
For encapsulation of hydrophilic drugs like rhodamine B - a water-soluble compound often used as a fluorescent probe to visualize and track drug carriers such as nanoparticles or micelles within cells and tissues - a double-emulsion w/o/w (water-in-oil-in-water) microfluidic method can be employed. In this setup, the inner aqueous phase containing Rhodamine B was securely encapsulated within a PLGA shell. This configuration ensures high encapsulation efficiency, enhanced stability, and extended drug release, making it not only ideal for both therapeutic and imaging applications, but also a representative model of microfluidic particle-based drug delivery.
Ongoing challenges and considerations in microfluidic drug-loaded PLGA
Microfluidics is revolutionizing drug encapsulation with precise control, high reproducibility, and innovations like stimuli-responsive particles and AI-driven optimization. However, PLGA-based encapsulation still faces key challenges that limit broader adoption and clinical translation.
Beyond formulation aspects, the fabrication of microfluidic chips is also critical, as variations in design or production methods can directly affect reproducibility and scalability.
Key challenges include:
- Solvent selection and removal processes must eliminate residual toxicity.
- Formulation must be optimized to prevent burst release and maintain stability.
- Large-scale production still requires careful engineering to retain uniformity at higher volumes.
Recent advances in microfluidic platforms – including compatibility with a broad range of organic solvents and surfactants – are helping to address these challenges and streamline formulation optimization. In parallel, progress in chip fabrication techniques is improving reproducibility and scalability, further supporting the translation toward clinical applications.
The future of drug delivery is here
The combination of microfluidic engineering, microfluidic encapsulation, and biodegradable polymers like PLGA is unlocking unprecedented control in drug delivery design - enabling more effective, safer, and patient-tailored therapies. The success of Risperdal Consta® demonstrates that PLGA encapsulation is not just a promising research tool, but a clinically validated strategy that can change lives. With microfluidics pushing particle precision and scalability even further, the next generation of PLGA-based systems could deliver similar breakthroughs for cancer treatment, chronic diseases, and beyond.
At Blacksheep Sciences, we’re helping biotech and pharmaceutical innovators harness this power with flexible, scalable microfluidic platforms.
Discover how our microfluidic platforms can advance your drug delivery goals at https://www.blacksheepsciences.com/resources



